Strategic Capability Building: Empowering Your Teams Through Targeted GMP Trainings.
A GMP system is not a static set of documents; it is a living culture. My training programs are designed to bridge the gap between regulatory requirements and operational reality. I focus on fostering deep understanding and personal ownership, ensuring that compliance becomes a seamless part of your daily practice.


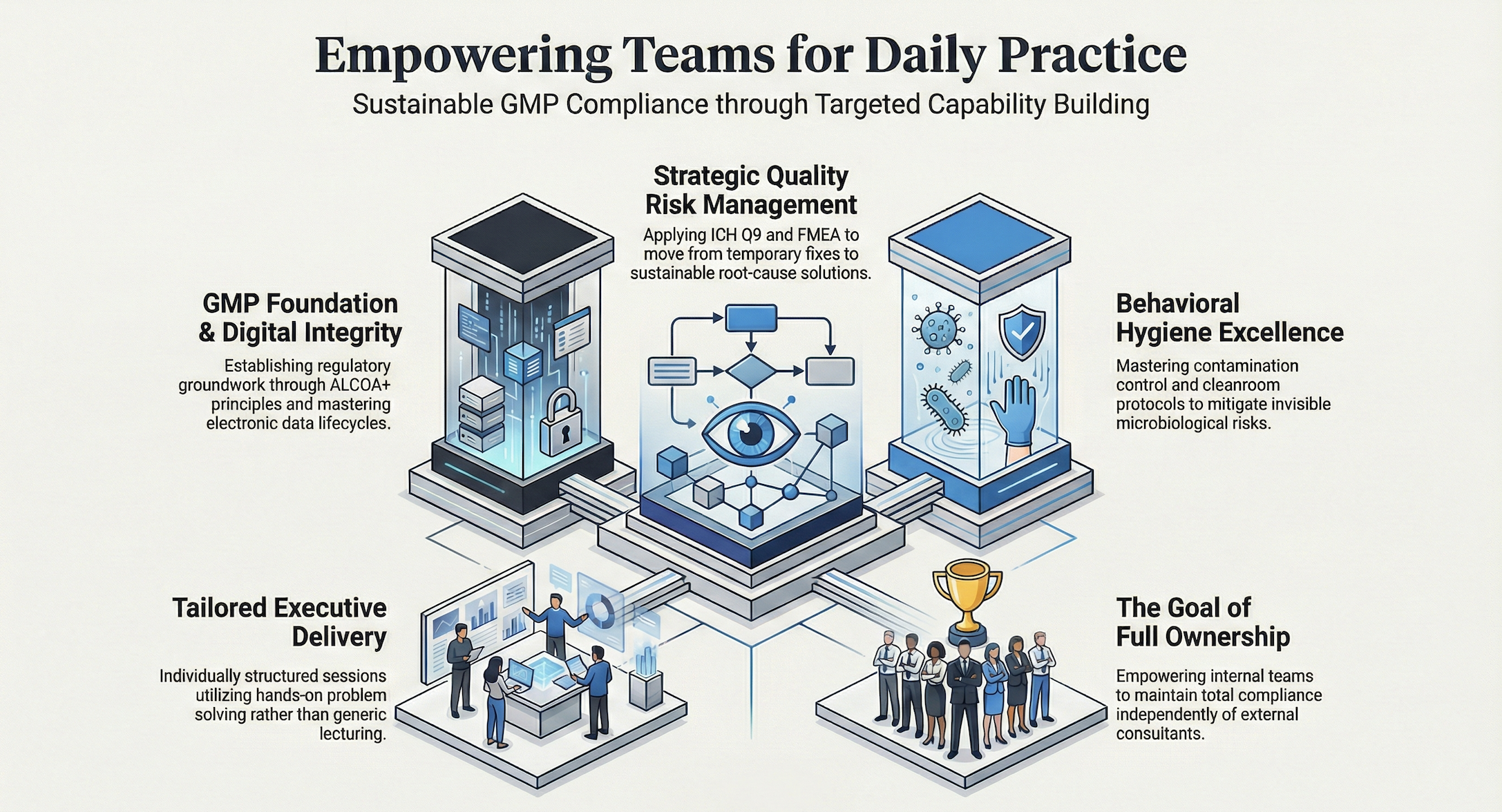
GMP Foundation & Excellence
Building the Regulatory Groundwork A robust quality culture begins with a shared understanding of the rules. These modules are designed to transform abstract regulations into a clear, actionable mindset for your team at every experience level.
GMP Basics for Beginners: An essential introduction to the world of Good Manufacturing Practices. This course provides newcomers with a solid understanding of the "Why" behind the regulations and their personal responsibility in the production chain.
GMP for Advanced Professionals: A high-level deep dive designed for experienced staff. We focus on complex regulatory updates, case studies from recent inspections, and the critical evaluation of established processes to prevent "compliance fatigue."
Digital Integrity & GDocP: Mastering the lifecycle of data. We bridge the gap between traditional Good Documentation Practice and modern Digital Integrity. This training ensures your team understands ALCOA+ principles in both paper-based and electronic systems, securing your data against unauthorized changes and ensuring full audit trail transparency.

Strategic Foresight & Operational Control
Strategic Foresight & Operational Control
Practical Risk Analysis: Moving beyond theory, we focus on the hands-on application of ICH Q9 and FMEA. Your team will learn how to identify, assess, and mitigate risks within your specific production environment to prevent systemic failures.
Deviation & CAPA Management: Turning obstacles into opportunities for improvement. We train your quality unit to handle OOS and OOT results effectively, ensuring that root cause investigations lead to sustainable solutions.
"Not sure where to start? Download my Training & Capability Statement (PDF) for an overview of all modules and my methodology."

Hygiene & Microbiology
Mastering the Invisible Challenges
Operational Hygiene & Cleanroom Behavior: Technical expertise is nothing without behavioral discipline. We focus on the human factor in Cleanroom Classes B–D, covering gowning procedures, material flow, and the prevention of cross-contamination.
Microbiology for Non-Microbiologists: Understanding the hidden risks. This training demystifies bioburden, endotoxins, and environmental monitoring, enabling your staff to recognize microbiological threats and respond with appropriate corrective actions.